Understanding Exoplanets’ Atmospheres in the Lab
Exoplanets are planets that orbit stars other than the Sun. These fascinating objects have only been observed in the last 30 years. Some exoplanets have atmospheres that can be detected and characterized using advanced technologies, such as the James Webb Space Telescope (JWST). Numerical simulation models have been developed to better understand and interpret observational data. Despite these major advances, however, the details known about these atmospheres remain limited. For example, we know that the atmosphere of K2-18 b, a planet located in the habitable zone of its star 124 light-years away, has a temperature similar to Earth’s. The atmosphere is dominated by hydrogen (H2), with a few percent of methane (CH4) and carbon dioxide (CO2). The exact concentrations of these compounds remain poorly understood, however, and the information we have on minority compounds is minimal. One way that scientists look to gain insight into exoplanetary atmospheres is by reproducing the atmospheres in laboratories.
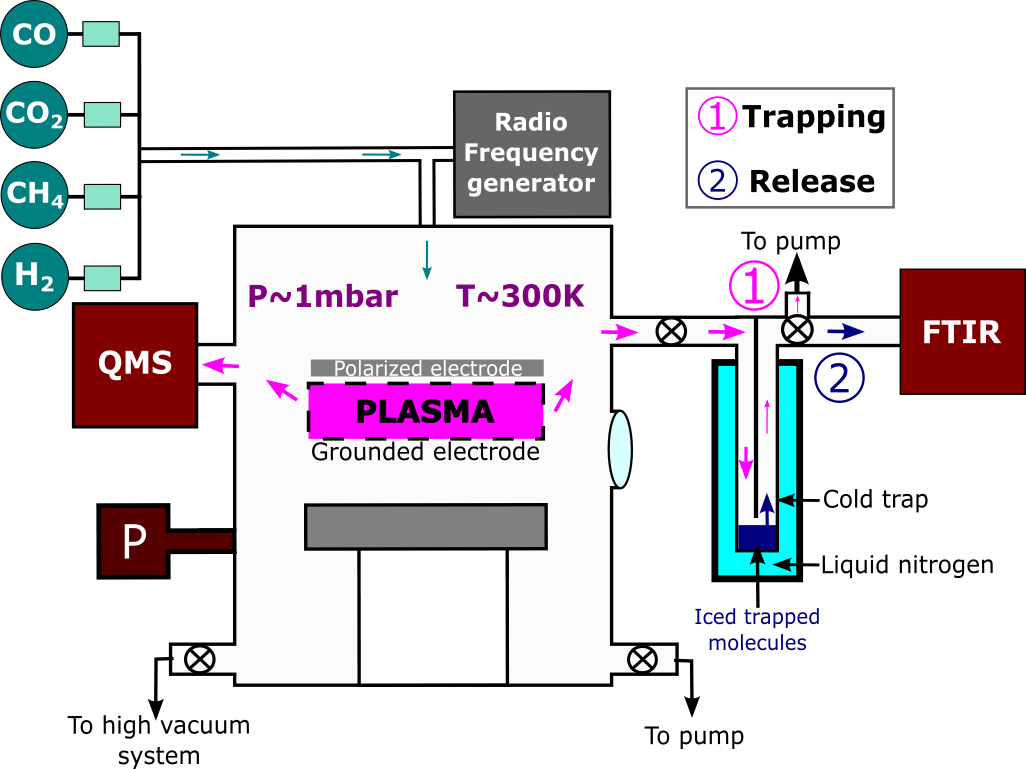
Sohier and their team produce analogues of exoplanetary atmospheres. To do this, they use an experimental setup consisting of a main chamber, into which gas mixtures are injected (Figure 1). These mixtures represent the atmospheres of exoplanets. The gas mixtures are then subjected to irradiation, which breaks the chemical equilibrium. This is much like when upper layers of atmospheres are exposed to high levels of UV radiation, and it is through these processes that the aurora borealis is formed.
During this irradiation, chemical bonds in the molecules are broken, and the atoms can reassemble to form new molecules. To observe what happens in the experiment, we use powerful analytical tools that allow us to see details that are invisible with JWST. O. Sohier uses two complementary analytical methods: mass spectrometry and infrared spectroscopy. Mass spectrometry is used to characterize the chemical structure of molecules by breaking them down into fragments whose masses are then measured. Each molecule thus has a mass spectrum that acts as its “fingerprint,” allowing it to be identified. Infrared spectroscopy is used to characterize the chemical bonds in molecules by measuring their absorption in the infrared domain. The complementary nature of these methods makes it possible to identify new chemical molecules as they form.
Figure 1: Scheme of the plasma reactor PAMPRE used to simulate the atmosphere of H₂-rich temperate exoplanets, at a pressure of 1 mbar and a temperature of 20°C. The tools used for analyses are QMS (Quadrupole Mass Spectrometer) and FTIR (Fourier Transform InfraRed spectrometer). P is a pressure gauge. A liquid nitrogen cold trap is used to condense the products in order to concentrate them and make them detectable.
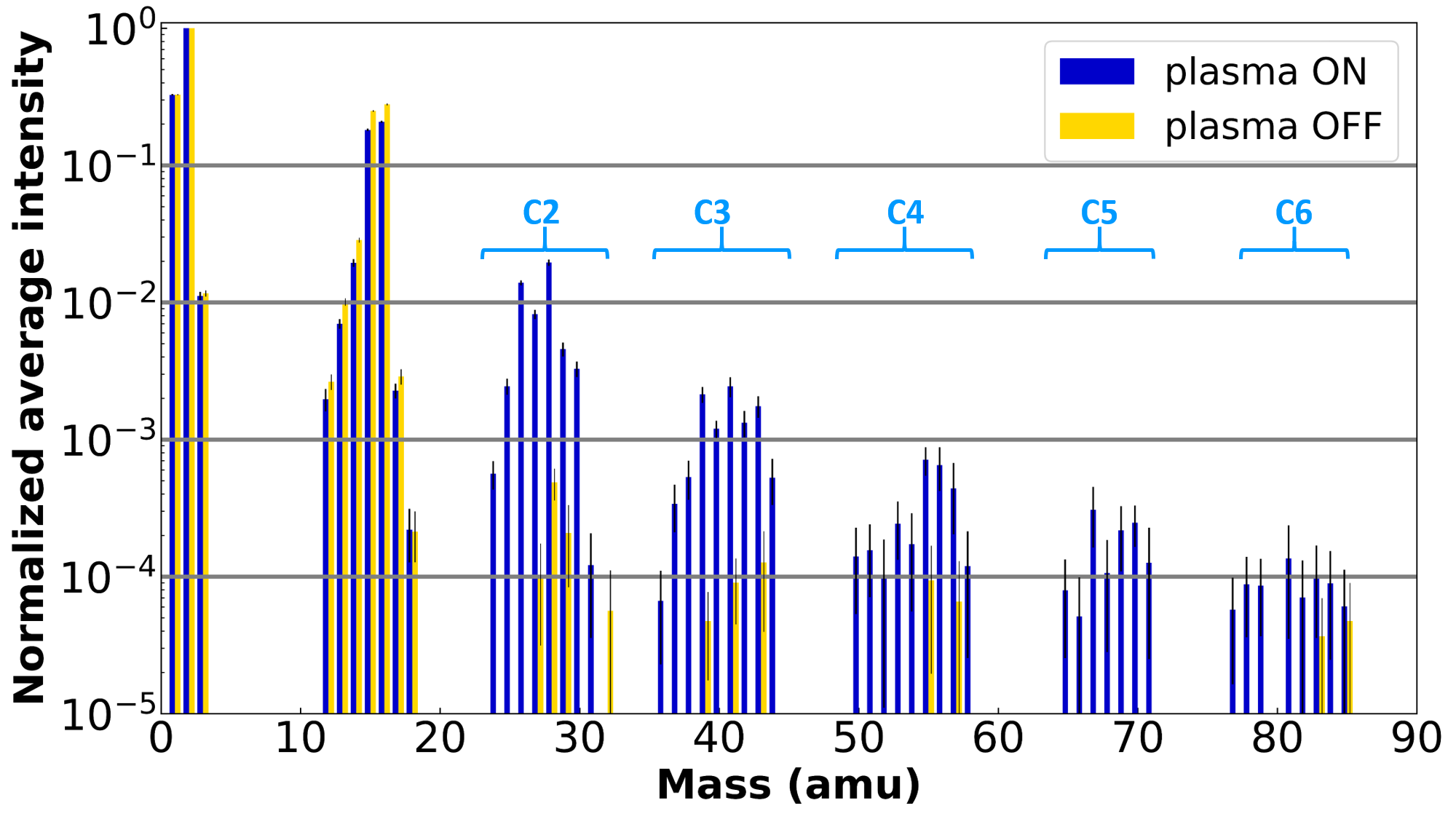
Sohier and their team have shown that relatively complex chemistry can take place in these atmospheres. They observe the formation of complex molecules, known as hydrocarbons, in atmospheres rich in CH4 (Figure 2). Hydrocarbons are chains containing several carbon atoms--up to six in this case--and hydrogen atoms. They also observe the formation of oxidized molecules in atmospheres rich in CO or CO2, which are known to be very important in prebiotic chemical reactions: formaldehyde (H₂CO), methanol (CH3OH), and acetaldehyde (CH3CHO). Using a numerical model, O. Sohier tries to understand how these molecules of interest form, and in what proportions, depending on different atmospheric compositions. This allows scientists to determine in which conditions molecules would be most detectable during real exoplanet observations. In CO2-rich atmospheres, few hydrocarbons and many oxidized organic compounds are formed. In atmospheres rich in both CH4 and CO, significant simultaneous formation of hydrocarbons and oxidized organic compounds is possible.
Figure 2: Mass spectrum (overlay of the specific “fingerprints” of all the molecules present) of a gas mixture of 95% H₂ + 5% CH4 before (gold) and during irradiation (blue). We observe the formation of peaks characteristic of carbon chains containing up to six carbon atoms, highlighted in light blue (C2 for two carbon atoms etc…).
This article made use of the following publication:
Sohier, O. et al., 2026, Astronomy & Astrophysics, 707, A53.
Original Contributor

O. Sohier
Laboratory for Atmospheres, Environments, Space Observations Laboratory
O. Sohier is a PhD student from the Laboratory for Atmospheres, Environments, Space Observations Laboratory (LATMOS) in Paris, working on the experimental simulation and characterization of exoplanetary atmospheres.
Editors
Katy Wetzel
Science Editor
Mélisse Bonfand
Science Editor
Annika Geiger
Senior Editor